Immunotoxicity Assays
The evaluation of the potential side effects of pharmaceuticals on the immune system should be incorporated into standard drug development process. Immunotoxicity refers to the toxic effects of xenobiotics, including pharmaceuticals, on the immune system's functioning. The immune system is a tightly regulated mechanism that mediates complex physiological responses to protect against invading pathogens and tumors. Toxicity to the immune system can result in unintended immunosuppression or stimulation (including hypersensitivity) of the immune response. Inhibiting the immune response may decrease host resistance to infectious agents or certain forms of nongenotoxic carcinogenicity, while enhancing it can exacerbate autoimmune diseases or allergies. Therefore, assessing whether pharmaceutical drugs negatively impact the immune system is an essential component of evaluating their safety.
Currently, the assessment of immunotoxic effects relies on various animal models. In vitro immunotoxicity testing offers several advantages over in vivo testing, including a comprehensive understanding of mechanisms, extrapolation across species (parallelogram approach), and the reduction, refinement, and replacement of animal experiments.
Following the FDA Guidance for Nonclinical Evaluation of the Immunotoxic Potential of Pharmaceuticals, PicoImmune has leveraged our expertise in immunology and flow cytometry to establish a set of standardized protocols for immunotoxicity testing that can effectively support drug discovery and development.
Immunotoxicity Testing
Humoral Immunity
- Serum globulin levels and albumin-to-globulin (A/G) ratios
- Immunoglobulin isotype distribution of IgM, IgG, IgA and IgE
- Immunoassay of serum cytokines
- Total hemolytic complement activity and components of complement
- Serum auto-antibody screening and deposition of immunoglobulins
Cellular Immunity
- Immunostaining and flow-cytometric analysis of B, T cells and subsets
- Mitogenic stimulation assays for B and T cells
- Natural killer (NK) cell activity
- Macrophage and neutrophil assays
- Mixed Lymphocyte Reaction
- Dendritic Cell Maturation
- Immunopathology
- Total leukocyte counts and absolute differential leukocyte counts
Hematotoxicity assays
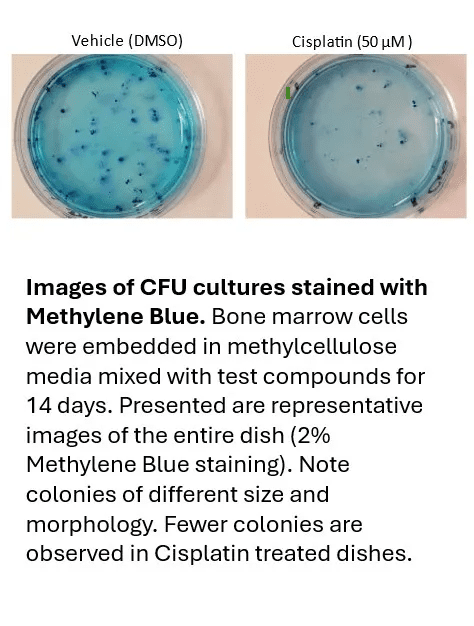
The colony forming cell (CFC) assay, also known as the methylcellulose assay, is an in vitro technique utilized in the investigation of hematopoietic stem cells. Hematopoietic Stem Cells (HSCs) are responsible for replenishing mature blood cells following hematopoietic stresses. Myelosuppression, which refers to adverse effects on the blood-forming system, is one of the most common dose-limiting side effects in cancer treatment. Understanding the potential hematotoxicity of candidate drugs is a crucial step in preclinical safety evaluation. The CFC assay relies on the capacity of hematopoietic stem and progenitor cells to proliferate and differentiate into colonies within a semi-solid medium upon cytokine stimulation. These formed colonies can be quantified and characterized based on their distinct morphology. The in vitro protocol involves treating mononuclear bone marrow cells or CD34+ cells with test drugs. This protocol offers a rapid quantitative assessment of any myelosuppressive potential exhibited by a test article.
Advantages of the CFU Assay:
- Provides clinically predictive information, enabling improved planning and reduced reliance on in vivo studies.
- Utilizes primary hematopoietic cells from various species (human, non-human primate, mouse, rat or dog) to aid in the selection of appropriate animal models.
- Evaluates both the proliferation and differentiation of erythroid (BFU-E) and myeloid (CFU-GM) progenitors.
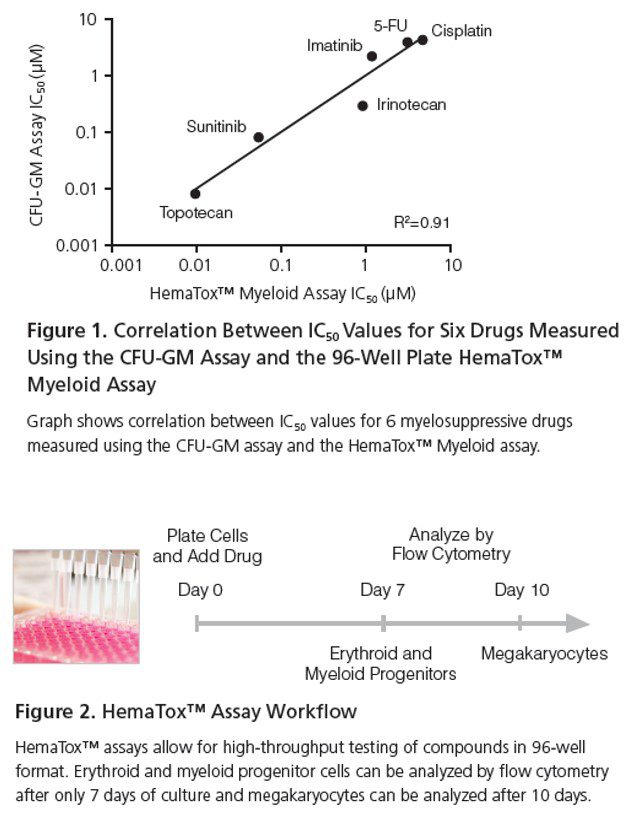
The HemaTox™ assays have been developed to assess the toxicity of drugs on the growth and lineage-specific differentiation of human CD34+ hematopoietic stem and progenitor cells (HSPCs) into one of three specific progenitor cell lineages, namely erythroid, myeloid, or megakaryocyte. These liquid medium-based assays demonstrate similar trends in drug toxicity as identified in CFU assays and can be conveniently conducted in a 96-well format. Moreover, the HemaTox™ assays offer flexibility in treatment regimens and enhance the ability to evaluate the effects of anti-proliferative drugs in vitro.
Advantages of HemaTox™ Liquid Medium-based Assays:
- Enables assessment of proliferation and differentiation of erythroid, myeloid, and megakaryocyte progenitors in < 10 days.
- Allows high-throughput testing of compounds in a 96-well format.
- Exhibits correlation with the CFU assay for toxicity levels across a wide range of compounds.
- Offers high flexibility as test compounds can be added to the culture at different time points, enabling examination of the effects on progenitor cells at various stages of differentiation.
- Provides improved sensitivity in evaluating anti-proliferative effects.
Genotoxicity assays
Genotoxic chemicals exert their adverse effect through interaction with DNA of cells. Genotoxicity tests are designed to detect drugs which can induce genetic damage directly or indirectly by various mechanisms of action. Compounds identified as genotoxic in these tests have the potential to be human carcinogens or mutagens, and ultimately may induce cancer or heritable defects.
Genotoxicity testing of new chemical entities is an integral part of the drug development process and is a regulatory requirement prior to the approval of new drugs. By identifying genotoxicity at an early stage in drug discovery rather than during regulatory assessment, the likelihood of late-stage failures is reduced.
PicoImmune offer a range of screening services to identify genotoxicity.
- Ames Test (OECD471): a standard test to assess mutagenic damage in a bacterial reverse gene mutation.
- COMET assay: a standard assay to access DNA damage, also known as single cell gel electrophoresis (damaged DNA image looks like a comet), is a sensitive test for analyzing DNA damage in individual cells.
- In vitro micronucleus test (MNT) (OECD487)
- In vitro chromosomal aberration test (OECD483)
- immunofluorescence staining of pH3 (phospho histone 3, a marker of G2/M phase)
- immunofluorescence staining of pH2AX (phospho histone 2AX, a marker of double strand DNA breaks)
- Other cell-based assays
